The treatment of tosylhydrazones with alkyl lithium reagents is called the Shapiro reaction. Tosyl hydrazone is a functional group with the general structure RRCN-NH-Ts, where Ts is a tosyl group. This reaction may be used to effect the overall transformation of a ketone to an alkene. While Bamford Stevens reaction yields more substituted olefins as the thermodynamic product (the product that predominates when the reaction is done at high temperature.) 2 3 4. The usage of aprotic solvent gives predominantly Z-alkenes, while protic solvent gives a mixture of E- and Z-alkenes. The Bamford–Stevens reaction is a chemical reaction whereby treatment of tosylhydrazones(p-Toluenesulfonyl hydrazones)with strong base gives alkenes.It is named for the British chemist William Randall Bamford and the Scottish chemist Thomas Stevens (1900–2000). Clicking on the donut icon will load a page at with additional details about the score and the social media presence for the given article. The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. The usage of aprotic solvents gives predominantly Z- alkenes, while protic solvent gives. 1 2 3 It is named for the British chemist William Randall Bamford and the Scottish chemist Thomas Stevens Stevens (19002000). Bamford-Stevens Reaction Shapiro Reaction W. Read 'ChemInform Abstract: SiliconDirected BamfordStevens Reaction of Trimethylsilyl N Aziridinylimines., ChemInform' on DeepDyve, the largest online rental service for scholarly research with thousands of academic publications available at your fingertips. These metrics are regularly updated to reflect usage leading up to the last few days. The BamfordStevens reaction is a chemical reaction whereby treatment of tosylhydrazones with strong base gives alkenes.
BAMFORD STEVENS REACTION PDF PDF
Learn more about these metrics Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. The advantage of the Shapiro over Bamford-Stevens Reaction is, that the resulting dianion does not tend to rearrange, which can. In aprotic conditions (sodium methoxide in.

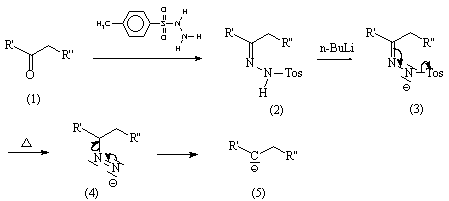
Bamford Biographical references: Campbell, W.A. (also in the Collective Volume (1990) 7:438 (PDF)) ^ Sarkar. The Bamford–Stevens reaction has not proved useful for the stereoselective generation of alkenes via.
